Review Article
Human Papillomavirus-Positive Oropharyngeal Squamous Cell Carcinoma Demographics, Prognosis, and Staging
Cleary RK and Cmelak AJ*
Department of Radiation Oncology, Vanderbilt University Medical Center, USA
*Corresponding author: Anthony J. Cmelak, Department of Radiation Oncology, Vanderbilt University Medical Center, Nashville, TN 37232-5671, USA
Published: 20 Jun, 2016
Cite this article as: Cleary RK, Cmelak AJ. Human Papillomavirus-Positive Oropharyngeal Squamous Cell Carcinoma Demographics, Prognosis, and Staging. Clin Oncol. 2016; 1: 1018.
Abstract
Human papillomavirus (HPV) is a sexually-transmitted infection that is responsible for increasing rates of oropharyngeal squamous cell carcinoma (OPSCC) in the United States and around the world. Compared to head and heck squamous cell carcinomas (HNSCC) caused by traditional risk factors such as smoking and alcohol, patients with HPV-positive (HPV+) OPSCC are younger, more likely to be male, and enjoy a significantly improved prognosis. As HPV status represents the most important prognostic variable for OPSCC patients, current staging systems which do not take it into account are inadequate for staging HPV+ OPSCC. Herein, we will review the shifting demographics of HPV infection as they relate to OPSCC incidence, outline the data suggesting improved outcomes in HPV+ OPSCC, and highlight current efforts to develop a new staging system for HPV+ OPSCC patients.
Introduction
Human papillomavirus (HPV) has been established as an increasingly important cause of oropharyngeal squamous cell carcinoma (OPSCC) worldwide by the WHO [1]. This understanding, as well as differences in prognosis, treatment response, and failure patterns, has led to a paradigm shift in our management of patients diagnosed with HPV-positive (HPV+) OPSCC. The large increase in HPV+ HNSCC and the associated improved response to treatment and overall survival have brought into question the validity of applying current staging systems to these patients as well as historical results from clinical trials that enrolled individuals with heterogeneous tumor sites in an era when the importance of HPV was not yet understood. Thus, a number of new efforts are under way to stratify patients based on HPV status, using multiple strategies to ameliorate treatment-related toxicities for HPV+ disease.
Demographics and Increasing Incidence of HPV Infection
An increase in OPSCC incidence rates has been observed worldwide over the past few decades,
including in the United States [2-6]. In order to examine the potential role for HPV as compared
to smoking on incidence trends, Chaturvedi et al. analyzed data regarding cancer incidence in the
Cancer Incidence in Five Continents database, a worldwide cancer registry. Using data collected
from 1983 to 2002, they compared incidence trends across multiple countries for traditionally
smoking-associated cancers, including lung and oral cavity squamous cell carcinoma (OCSCC),
versus OPSCC. In general, OPSCC incidence rates were noted to increase significantly, primarily in
developed countries, despite concomitant decreases in OCSCC and lung cancer incidence in many
of the same countries. Men were disproportionately affected as compared to women, and young
men (<60 yo) experienced higher rates of OPSCC than older men. Furthermore, incidence rates
of all three cancer types were noted to increase in women. Taken together, these suggest a role for
HPV infection in the increasing incidence of OPSCC, especially among men, with smoking being
the primary driver for increased rates of these cancers among women.
A preponderance of data exists to demonstrate the role of HPV in increasing OPSCC incidence
in the United States and abroad [4,7]. Several series have used molecular testing to show increased
rates of HPV+ OPSCC while rates of HPV-negative (HPV−) OPSCC continue to decline. Further
underscoring this point, a recent meta-analysis of 2,099 OPSCC cases from the US literature showed
an increase in the prevalence of HPV+ OPSCC from 20.9% before 1990 to 51.4% between 1990-
1999 with further increase to 65.4% for 2000-present [8]. Additional series suggest 72% or more of
OPSCC cases occurring in the United States after the year 2000 are attributable to HPV infection
[7,9]. Impressively, population level incidence of HPV+ OPSCC was shown to increase by 225%
from 1988 to 2004 while incidence of HPV− cancers declined by 50% (Table 1).
The primary risk factor for development of HPV+ OPSCC is
oral HPV infection, most frequently by HPV type 16 [10,11]. As the
vast majority of oral HPV infections are sexually acquired, sexual
behavior has now been established as a risk factor for HNSCC,
specifically OPSCC [10,12]. Lifetime number of oral sexual partners
is the behavior with the strongest association with OPSCC incidence
[13]. The reported rates of engaging in oral sex vary significantly on
a geographic basis as well as across birth cohorts, with more recent
cohorts reporting significantly higher rates of oral sex [13]. These
may help explain the heterogeneous incidence of HPV+ OPSCC
worldwide as well as the increasing incidence over time.
In the United States, men have a significantly higher prevalence
of overall oral HPV infection (10.1% vs. 3.6%) and oral HPV type
16 infection (1.6% vs. 0.3%) than women, offering an explanation
for the gender inequality of OPSCC incidence [12]. The difference
in incidence is likely multifactorial as men report a higher number
of lifetime sexual partners than women [14], have a higher perpartner
increase in risk of high risk oral HPV infection [15], and
are less likely to seroconvert after genital HPV infection to provide
protection against subsequent oral HPV infection [15]. Prevalence
of oral oncogenic HPV infection in the United States has a bimodal
distribution with peaks noted between the ages of 25 to 30 years
and 55 to 60 years [16], while the median age at diagnosis of HPV+ OPSCC is 58 years [17]. This suggests a latency period of 5-30 years as
uncertainty exists regarding which of the two peaks plays the greatest
role in HPV-related oncogenesis. Interestingly, long-term partners of
HPV+ OPSCC patients do not appear to have elevated oral oncogenic
HPV infection rates compared to the general population (1.2% vs.
1.3%) in spite of high prevalence of oncogenic oral HPV in the
patients themselves, offering reassurance to partners regarding their
risk of developing OPSCC.
Table 1
Table 1
Population level incidence of HPV-positive OPSCC was shown to increase by 225% from 1988 to 2004.
From: Chem Res Toxicol. 2014 Apr 21; 27(4): 462–469. Published online 2014 Mar 18. doi: 10.1021/tx500034c.
Figure 1
Figure 1
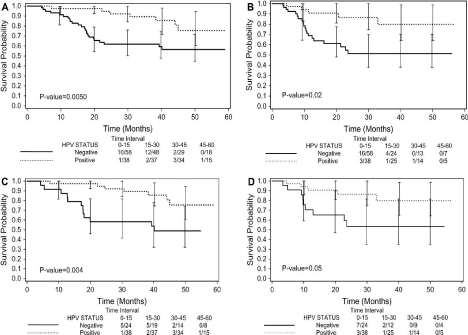
Kaplan–Meier curves for overall and progression-free survival stratified by tumor human papillomavirus (HPV) status.
Kaplan–Meier curves for overall and progression-free survival stratified by tumor human papillomavirus (HPV) status. A) Overall survival (OS) for the entire study
population. B) Progression-free survival (PFS) for the entire study population. C) OS for patients with oropharynx cancer only. D) PFS for patients with oropharynx
cancer only. From: Carole Fakhry et al. JNCI J Natl Cancer Inst. 2008; 100: 261-269. (Oxford university Press).
HPV and OPSCC: Prognostic Implications
[19-22] initially
suggested this difference in outcomes, it was first confirmed in a
prospective manner by Fakhry et al. [23] through analysis of the results
of ECOG 2399 [24]. This Phase II trial included 105 resectable
stage III/IVA larynx and OPSCC patients treated with two cycles
of induction carboplatin and paclitaxel followed by concurrent
paclitaxel and radiation to 70 Gy if no evidence of tumor progression
was observed. HPV status was tested prospectively in 96 patients
using polymerase chain reaction (PCR) and in situ hybridization
(ISH) with 40% of patients harboring oncogenic HPV DNA. Higher
response rates to induction chemotherapy (IC) (84% vs. 55%) and
chemoradiotherapy (CRT) (84% vs. 57%) were noted among patients
with HPV+ versus HPV− tumors, respectively, as well as improved
2-year overall survival (OS) (95% vs. 62%) (Figure 1).
Bonner et al. [25] performed a phase III randomized study of 424
patients with locally-advanced HNSCC comparing radiotherapy
alone with radiotherapy plus cetuximab with updated 5-year results
confirming improved locoregional control (LRC), progression-free
survival (PFS), and OS in the combined modality group [26]. A
retrospective analysis of 182 patients from the trial with OPSCC and
evaluable HPV status via IHC confirmed these improved outcomes
in both the HPV+ and HPV− OPSCC patient subsets suggesting
no predictive ability of HPV-status for response to cetuximab [27].
Additionally, improvements in LRC, PFS, and OS were noted in the
HPV+ versus HPV− OPSCC patient populations, again confirming
the prognostic role of HPV.
The Radiation Therapy Oncology Group (RTOG) 0129 trial
included a total of 743 patients with stage III/ IV HNSCC randomized
to concurrent cisplatin plus accelerated fractionation radiation with
a concomitant boost versus standard fractionation radiation [28].
With 8 years of follow-up, no OS difference was seen between the two
radiation fractionation regimens. However, the study did confirm
a survival difference between HPV+ and HPV− OPSCC regardless
of treatment modality at 8 years (71% vs. 30%, respectively). A
retrospective analysis performed by Ang et al. [29]identified the dominant
prognostic factors predictive of overall survival in the 323 OPSCC
patients on trial tested for HPV which included HPV status, packyears
of tobacco smoking, tumor stage, and nodal stage.
Importantly, smoking history was shown to abrogate the beneficial
effects of HPV status as the risks of cancer death or relapse were
increased by 1% for each additional year of tobacco smoking in both
HPV− and HPV+ cohorts. A cut-off point of 10 pack-years was found
to be the best predictor of survival related to smoking status. Using
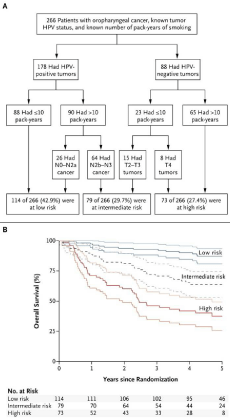
a recursive partitioning analysis (RPA), patients were classified as
having a low, intermediate, or high risk of death based on these four
factors with 3-year survival rates of 93%, 71%, and 46%, respectively
(Figure 2).
A retrospective analysis of 505 patients with OPSCC and
evaluable HPV status treated with definitive radiation (RT) or CRT
between 2001 to 2009 was performed by O'Sullivan et al.[30] in order to
identify subgroups of patients suitable for treatment deintensification
based on low risk of distant metastasis. HPV+ patients were
noted to have improved local (94% vs. 80%) and regional (95% vs.
82%) control versus HPV− patients, though similar distant control
(90% vs. 86%) was found. Smoking pack-years >10 was confirmed
to be associated with reduced overall survival. RPA was performed
to segregate patients into low-risk (T1-3, N0-2c) and high-risk (T4
or N3) groups for distant metastasis with distant control of 93%
and 76%, respectively. Notably, distant control rates were similar
for HPV+, low-risk N0-2a patients or less than 10 pack-year N2b
patients regardless of treatment with RT or CRT but were worse for
HPV+ N2c patients managed with RT alone versus CRT (73% vs.
92%, respectively), indicating that this patient subset is not ideallysuited
for chemotherapy de-intensification.
Figure 2
Figure 2
Classification of the study patients into risk-of-death categories
and kaplan–meier estimates of overall survival according to those categories.
From: Ang KK et al. N Engl J Med. 2010; 363: 24-35.
Table 2
Table 2
Proposed ICON-S stage tabulation grid for 8th edition TNM Note that
distant metastatic disease (M1) is considered stage IV.
From: O’Sullivan B, et.al. Lancet Oncol. 2016 Feb 26. pii: S1470-2045(15)00560-
4. doi: 10.1016/S1470-2045(15)00560-4.
HPV and OPSCC Staging Systems
As HPV status has become the most important prognostic
variable for OPSCC patients, efforts are underway to refine the current
traditional TNM staging system defined by the American Joint
Committee on Cancer (AJCC) which does not account for HPV [31].
Significant stage migration has occurred over time with the increasing
incidence of HPV+ OPSCC as these patients typically present with
a lower T stage and higher N stage than their HPV− counterparts
[32,33]. Unsurprisingly, the AJCC TNM stage is no longer prognostic
for HPV+ patients, though it does retain prognostic capacity for
HPV− patients [32-35]. Underscoring the point, improved survival
has been observed in OPSCC patients in the United States with N2a
versus N0 nodal stage, ostensibly due to the frequency of lymph node
(LN) metastasis with HPV+ patients [36,37].
Several groups have sought to refine the current AJCC staging
system for non-metastatic HPV+ OPSCC. Huang et al. [35] performed an
analysis of 573 HPV+ OPSCC patients treated at Princess Margaret
from 2000 to 2010 using RPA to derive prognostic groups. Their
initial RPA defined risk groups based solely on AJCC T and N stage
classifications and divided patients into RPA-I (T1-3N0-2b), RPA-II
(T1-3N2c), and RPA-III (T4 or N3) groups with 5-year OS of 82% vs.
76% vs. 54%, respectively. An additional RPA which included age and
smoking pack-years in addition to RPA stage further segregated the
cohort into four prognostic groups: group I (T1-3N0-N2c, <20 packyears),
group II (T1-3N0-2c, > 20 pack-years), group III (T4 or N3,
age < 70), and group IV (T4 or N3, age >70) with 5-year OS 89%, 64%,
57%, and 40%, respectively. The model was validated internally, but
external validation was not performed as part of the analysis.
Using a separate cohort of 662 HPV+ OPSCC patients treated at
MD Anderson between 2003 and 2012, Dhalstrom et al. were unable
to validate the stage and prognostic groups proposed by Huang et al.
[38]. They proposed their own staging system using nasopharyngeal
(NPC) N categories rather than the traditional OPSCC regional
LN categories citing the similarity between NPC and HPV-related
OPSCC in regards to their viral causation and different natural
history versus other HNSCC subsites. RPA stratified patients into the
following stages: stage IA (T1, N0-2), stage IB (T2, N0-2), stage III
(T1-3, N3), stage IV (T4, Any N). 5-year OS for the risk groups were
94%, 87%, 76%, and 69%, respectively. Though smoking history (<
10 vs. >10 pack-years) was predictive for OS for the whole cohort
of patients, it was not able to stratify patient outcomes within each
stage group. Though the two models differ significantly, they do share
tumor volume as measured by T stage as an important prognostic
variable. This finding confirms observations made by others regarding
the prognostic importance of T stage versus N stage in HPV+ OPSCC
[33,34,39].
The International Collaboration on Oropharyngeal cancer
Network for Staging (ICON-S) sought to further refine and validate
the work undertaken by Princess Margaret to develop a TNM
classification system specific to HPV+ OPSCC in a multi-institutional
cohort study involving 7 centers across Europe and North America
[40]. The original training cohort from Princess Margaret was
expanded to 661 patients with further follow-up time, and 1246
patients were enrolled in a validation cohort at 6 additional centers
for a total of 1907 patients. Definitive treatment for the vast majority
of patients (98%) was non-surgical. RPA and adjusted hazard ratio
(AHR) modeling were used to create new staging classifications
for HPV+ OPSCC in the training cohort. These were subsequently
verified in the validation cohort. As noted previously, similar 5-year
OS was seen for AJCC 7th edition stage I, II, III, and IVa patients,
though stage IVb did prove to have a worse survival. Patients in
the AJCC N0, N1-N2a, and N2b subsets did not have a difference
in 5-year OS, though survival was significantly lower for those with
N3 disease. AHR modeling produced the TNM stage classification
that was most predictive of survival. As patients classified with
T4a and T4b disease experienced similar 5-year OS, T4 was no
longer subdivided in the new ICON-S T stage classification. Given
the similar prognosis for N1, N2a, and N2b patients, the following
ICON-S N stage classifications were proposed which closely parallel
those for NPC: ICON-S N0 (no LN); ICON-S N1 (ipsilateral LN);
ICON-S N2 (bilateral or contralateral LN); ICON-S N3 (LN > 6 cm).
The proposed ICON-S TNM staging classifications were as follows:
stage I (T1-2, N0-1); stage II (T1-2 N2, or T3N0-N2); stage III (T4
or N3); stage IV (M1). These closely parallel the initial findings from
Huang et al. except in the case of T3N0-N2b which is considered
ICON-S stage II rather than stage I. Heterogeneity tests showed that
HRs between the proposed stage groups were consistent across all
institutions, suggesting wide applicability of the staging system across
varied patient populations. In an exploratory training cohort of 702
patients, the additional risk factors of lower neck LN involvement
and > 5 positive LNs were investigated. Lower neck involvement
did appear to be associated with worse survival, but his was not an
independent effect and was left out of the final staging model. As
with lower neck involvement, > 5 LNs did not predict for survival
across all cohorts and was omitted from the staging system. Notably,
though smoking status was used as a variable to derive the prognostic
groupings, it was not included as a staging variable as the authors
wished to derive an anatomical stage classification (Table 2).
Conclusion
HPV+ OPSCC is a separate disease entity from HPV− HNSCC that increasingly impacts younger, healthier patients and portends a markedly improved prognosis. Further work is needed to establish staging systems that accurately stratify OPSCC based on outcomes. This will aid current efforts at treatment modification based on HPV status which aim to de-intensify therapy in HPV+ patients in order to reduce the long-term toxicities of treatment while escalating therapy in HPV− patients.
References
- International Agency for Research on Cancer ed. IARC monographs on the evaluation of carcinogenic risks to humans, volume 90, Human papillomaviruses: this publication represents the views and expert opinions of an IARC Working Group on the Evaluation of Carcinogenic Risks to Humans, which met in Lyon, 15 - 22 February 2005. Lyon: IARC; 2007.
- Reddy VM, Cundall-Curry D, Bridger MW. Trends in the incidence rates of tonsil and base of tongue cancer in England, 1985-2006. Ann R Coll Surg Engl. 2010; 92: 655-659.
- Jemal A, Simard EP, Dorell C, Noone AM, Markowitz LE, Kohler B, et al. Annual Report to the Nation on the Status of Cancer, 1975-2009, featuring the burden and trends in human papillomavirus (HPV)-associated cancers and HPV vaccination coverage levels. J Natl Cancer Inst. 2013; 105: 175- 201.
- Hammarstedt L, Lindquist D, Dahlstrand H, Romanitan M, Dahlgren LO, Joneberg J, et al. Human papillomavirus as a risk factor for the increase in incidence of tonsillar cancer. Int J Cancer. 2006; 119: 2620-2623.
- Forte T, Niu J, Lockwood GA, Bryant HE. Incidence trends in head and neck cancers and human papillomavirus (HPV)-associated oropharyngeal cancer in Canada, 1992-2009. Cancer Causes Control. 2012; 23: 1343-1348.
- Blomberg M, Nielsen A, Munk C, Kjaer SK. Trends in head and neck cancer incidence in Denmark, 1978-2007: focus on human papillomavirus associated sites. Int J Cancer. 2011; 129: 733-741.
- Chaturvedi AK, Engels EA, Pfeiffer RM, Hernandez BY, Xiao W, Kim E, et al. Human papillomavirus and rising oropharyngeal cancer incidence in the United States. J Clin Oncol. 2011; 29: 4294-4301.
- Stein AP, Saha S, Yu M, Kimple RJ, Lambert PF. Prevalence of human papillomavirus in oropharyngeal squamous cell carcinoma in the United States across time. Chem Res Toxicol. 2014; 27: 462-469.
- Steinau M, Saraiya M, Goodman MT, Peters ES, Watson M, Cleveland JL, et al. Human papillomavirus prevalence in oropharyngeal cancer before vaccine introduction, United States. Emerg Infect Dis. 2014; 20: 822–828.
- Gillison ML, Alemany L, Snijders PJ, Chaturvedi A, Steinberg BM, Schwartz S, et al. Human papillomavirus and diseases of the upper airway: head and neck cancer and respiratory papillomatosis. Vaccine. 2012; 30: F34-54.
- Gillison ML, D'Souza G, Westra W, Sugar E, Xiao W, Begum S, et al. Distinct risk factor profiles for human papillomavirus type 16-positive and human papillomavirus type 16-negative head and neck cancers. J Natl Cancer Inst. 2008; 100: 407-420
- Gillison ML, Broutian T, Pickard RK, Tong ZY, Xiao W, Kahle L, et al. Prevalence of oral HPV infection in the United States, 2009-2010. JAMA. 2012; 307: 693-703.
- Heck JE, Berthiller J, Vaccarella S, Winn DM, Smith EM, Shan'gina O, et al. Sexual behaviours and the risk of head and neck cancers: a pooled analysis in the International Head and Neck Cancer Epidemiology (INHANCE) consortium. Int J Epidemiol. 2010; 39: 166-181.
- D'Souza G, Cullen K, Bowie J, Thorpe R, Fakhry C. Differences in oral sexual behaviors by gender, age, and race explain observed differences in prevalence of oral human papillomavirus infection. PLoS One. 2014; 9: e86023.
- Giuliano AR, Nyitray AG, Kreimer AR, Pierce Campbell CM, Goodman MT, Sudenga SL, et al. EUROGIN 2014 roadmap: differences in human papillomavirus infection natural history, transmission and human papillomavirus-related cancer incidence by gender and anatomic site of infection. Int J Cancer. 2015; 136: 2752–2760.
- Chaturvedi AK, Engels EA, Anderson WF, Gillison ML. Incidence trends for human papillomavirus-related and -unrelated oral squamous cell carcinomas in the United States. J Clin Oncol. 2008; 26: 612-619.
- Chaturvedi AK. Tonsillectomy and Risk of Oropharyngeal Cancer: Implications for Research and Prevention. Cancer Prev Res (Phila). 2015; 8: 577-579.
- D'Souza G, Gross ND, Pai SI, Haddad R, Anderson KS, Rajan S, et al. Oral human papillomavirus (HPV) infection in HPV-positive patients with oropharyngeal cancer and their partners. J Clin Oncol. 2014; 32: 2408- 2415.
- Mehta V, Yu GP, Schantz SP. Population-based analysis of oral and oropharyngeal carcinoma: changing trends of histopathologic differentiation, survival and patient demographics. Laryngoscope. 2010; 120: 2203-2212.
- Andl T, Kahn T, Pfuhl A, Nicola T, Erber R, Conradt C, et al. Etiological involvement of oncogenic human papillomavirus in tonsillar squamous cell carcinomas lacking retinoblastoma cell cycle control. Cancer Res. 1998; 58: 5-13.
- Mellin H, Friesland S, Lewensohn R, Dalianis T, Munck - Wikland E. Human papillomavirus (HPV) DNA in tonsillar cancer: clinical correlates, risk of relapse, and survival. Int J Cancer. 2000; 89: 300-304.
- Ragin CC, Taioli E. Survival of squamous cell carcinoma of the head and neck in relation to human papillomavirus infection: review and metaanalysis. Int J Cancer. 2007; 121: 1813-1820.
- Fakhry C, Westra WH, Li S, Cmelak A, Ridge JA, Pinto H, et al. Improved survival of patients with human papillomavirus-positive head and neck squamous cell carcinoma in a prospective clinical trial. J Natl Cancer Inst. 2008; 100: 261-269.
- Cmelak AJ, Li S, Goldwasser MA, Murphy B, Cannon M, Pinto H, et al. Phase II trial of chemoradiation for organ preservation in resectable stage III or IV squamous cell carcinomas of the larynx or oropharynx: results of Eastern Cooperative Oncology Group Study E2399. J Clin Oncol. 2007; 25: 3971–3977.
- Bonner JA, Harari PM, Giralt J, Cohen RB, Jones CU, Sur RK, et al. Radiotherapy plus cetuximab for locoregionally advanced head and neck cancer: 5-year survival data from a phase 3 randomised trial, and relation between cetuximab-induced rash and survival. Lancet Oncol. 2010; 11: 21–28.
- Bonner JA, Harari PM, Giralt J, Azarnia N, Shin DM, Cohen RB, et al. Radiotherapy plus cetuximab for squamous-cell carcinoma of the head and neck. N Engl J Med. 2006; 354: 567-578.
- Rosenthal DI, Harari PM, Giralt J, Bell D, Raben D, Liu J, et al. Association of Human Papillomavirus and p16 Status With Outcomes in the IMCL- 9815 Phase III Registration Trial for Patients With Locoregionally Advanced Oropharyngeal Squamous Cell Carcinoma of the Head and Neck Treated With Radiotherapy With or Without Cetuximab. J Clin Oncol. 2016; 34: 1300-1308.
- Nguyen-Tan PF, Zhang Q, Ang KK, Weber RS, Rosenthal DI, Soulieres D, et al. Randomized phase III trial to test accelerated versus standard fractionation in combination with concurrent cisplatin for head and neck carcinomas in the Radiation Therapy Oncology Group 0129 trial: longterm report of efficacy and toxicity. J Clin Oncol. 2014; 32: 3858–3866.
- Ang KK, Harris J, Wheeler R, Weber R, Rosenthal DI, Nguyen-Tân PF, et al. Human papillomavirus and survival of patients with oropharyngeal cancer. N Engl J Med. 2010; 363: 24-35
- O'Sullivan B, Huang SH, Siu LL, Waldron J, Zhao H, Perez-Ordonez B, et al. Deintensification candidate subgroups in human papillomavirusrelated oropharyngeal cancer according to minimal risk of distant metastasis. J Clin Oncol. 2013; 31: 543–550.
- Edge SB, Compton CC. The American Joint Committee on Cancer: the 7th edition of the AJCC cancer staging manual and the future of TNM. Ann Surg Oncol. 2010; 17: 1471-1474.
- Dahlstrom KR, Calzada G, Hanby JD, Garden AS, Glisson BS, Li G, et al. An evolution in demographics, treatment, and outcomes of oropharyngeal cancer at a major cancer center: a staging system in need of repair. Cancer. 2013; 119: 81–89.
- Keane FK, Chen YH, Neville BA, Tishler RB, Schoenfeld JD, Catalano PJ, et al. Changing prognostic significance of tumor stage and nodal stage in patients with squamous cell carcinoma of the oropharynx in the human papillomavirus era. Cancer. 2015; 121: 2594-2602.
- Ward MJ, Mellows T, Harris S, Webb A, Patel NN, Cox HJ, et al. Staging and treatment of oropharyngeal cancer in the human papillomavirus era. Head Neck. 2015; 37: 1002-1013.
- Huang SH, Xu W, Waldron J, Siu L, Shen X, Tong L, et al. Refining American Joint Committee on Cancer/Union for International Cancer Control TNM stage and prognostic groups for human papillomavirusrelated oropharyngeal carcinomas. J Clin Oncol. 2015; 33: 836–845.
- Fritsch VA, Sharma AK, Shirai K, Day TA. OP024: Improved survival for N2A oropharynx squamous cell carcinoma vs. N0/N1: Is our staging system still accurate? A population-based analysis of 15,588 cases. Oral Oncol. 2013; 49: S12–S13.
- Rios Velazquez E, Hoebers F, Aerts HJWL, Rietbergen MM, Brakenhoff RH, Leemans RC, et al. Externally validated HPV-based prognostic nomogram for oropharyngeal carcinoma patients yields more accurate predictions than TNM staging. Radiother Oncol. 2014; 113: 324–330.
- Dahlstrom KR, Garden AS, William WN Jr, Lim MY, Sturgis EM. Proposed Staging System for Patients With HPV-Related Oropharyngeal Cancer Based on Nasopharyngeal Cancer N Categories. J Clin Oncol. 2016; 34: 1848-1854.
- Klozar J, Koslabova E, Kratochvil V, Salakova M, Tachezy R. Nodal status is not a prognostic factor in patients with HPV-positive oral/oropharyngeal tumors. J Surg Oncol. 2013; 107: 625-633.
- O'Sullivan B, Huang SH, Su J, Garden AS, Sturgis EM, Dahlstrom K, et al. Development and validation of a staging system for HPVrelated oropharyngeal cancer by the International Collaboration on Oropharyngeal cancer Network for Staging (ICON-S): a multicentre cohort study. Lancet Oncol. 2016; 17: 440–451.